Ghasem Zolfaghari
Abstract
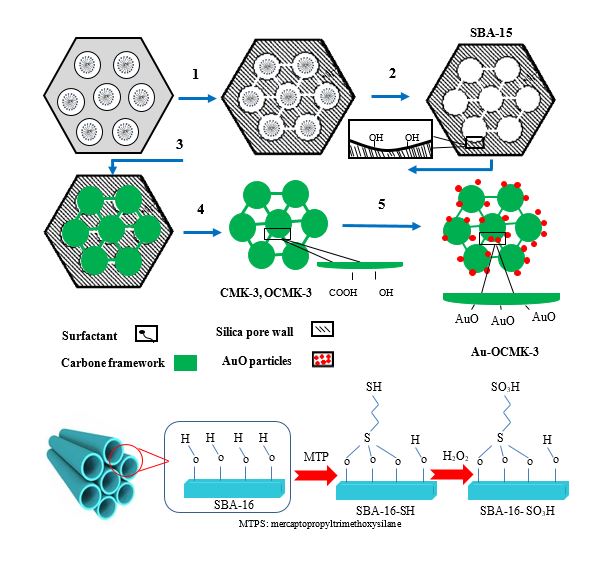
At present study, the removal of p-nitrophenol (PNP) by a newly designed mesoporous organocarbon, monolayers of ß-cyclodextrine (CD) on oxidzed ordered nanoporous carbon (OX-ONC) via 1,4-phenylene diisocyanate (PDI) linking denoted as CD-ONC was optimized. Furthermore, Au-doped mesoporous carbon ...
Read More
At present study, the removal of p-nitrophenol (PNP) by a newly designed mesoporous organocarbon, monolayers of ß-cyclodextrine (CD) on oxidzed ordered nanoporous carbon (OX-ONC) via 1,4-phenylene diisocyanate (PDI) linking denoted as CD-ONC was optimized. Furthermore, Au-doped mesoporous carbon CMK-3 denoted as Au-OCMK-3 was synthesized by using SBA-15. Au-OCMK-3 has been studied for removal of dibenzothiphene (DBT) and carbazole (CA) from n-hexane. Also the functionalization of SBA-16 mesoporous with sulfonic acid for arsenic (As (V)) and copper (Cu (II)) removal were carried out (SBA-16-SO3H). Maximum absorption capacity of CD-ONC was 100 mg/g. Dubinin–Radushkevich isotherm was applied to describe the nature of PNP uptake and it was found that it occurred physically (E = 0.07 KJ/mol, CD-ONC). Value for Temkin’s heat of adsorption is positive for PNP (157.87 J/mol, CD-ONC). There are two physisorption models of PNP with the surface C=O groups of ONC (H-bond and dispersion effect between phenolic ring and π electrons). The overall PNP adsorption process was exothermic and spontaneous in nature according to thermodynamics parameters (free energy (ΔGo), enthalpy (ΔHo), and entropy (ΔSo)). We demonstrate that functionalization of CMK-3 with gold is possible (qm value for DBT: 15.33 mg/g and for CA: 13.00 mg/g). The adsorption capacity for As (V) on SBA-16- SO3H reaches 92.63 mg/g. The high removal of As equilibrium time of 90 minutes can be explained in terms of a strong electrostatic attraction that occurred between the SO3H and As. Maximum absorption capacity was 92.63 mg/g for As(V) was and 13.00 mg/g for Cu(II).
Ashwini Prabhakar Shende; NILANJANA MITRA
Abstract
In recent years, the development in the field of nanotechnology is due to the fascinating properties of nanoparticles. In the present study, plant based bioflocculant extracted from the fruits of Okra (Abelmoschus esculentus) was purified, characterized and used for the biosynthesis of iron nanoparticles. ...
Read More
In recent years, the development in the field of nanotechnology is due to the fascinating properties of nanoparticles. In the present study, plant based bioflocculant extracted from the fruits of Okra (Abelmoschus esculentus) was purified, characterized and used for the biosynthesis of iron nanoparticles. Fourier transform infra-red (FT-IR) spectral analysis revealed the presence of hydroxyl, carboxyl and sugar derivatives in the bioflocculant. The biosynthesized Fe nanoparticles were characterized using UV-vis spectroscopy, X-ray diffraction (XRD), Fourier transform infra-red (FT-IR), Scanning electron microscopy (SEM) and Atomic force microscopy (AFM). TEM analysis was performed and the size of synthesized Fe nanoparticles was found to be 50 nm which was assessed by dynamic light scattering (DLS) analysis. Flocculation activity of bioflocculant mediated Fe nanoparticles (BFFeNPs) was tested. The effects of various parameters on Pb(II)removal using BFFeNPs were evaluated using response surface methodology (RSM) based on Box Behnken Design (BBD).The BFFeNPs exhibited high Pb (II) removal efficiency (91.45%) under optimized parameters viz. pH 6, BFFeNPs dosage 0.2 g/L, contact time 30 min and temperature 30º C. A quadratic polynomial model was fit with the actual data of R2 0.99 for metal removal. To the best of our knowledge, this is the first report on the potential use of Okra bioflocculant mediated Fe nanoparticles synthesis for the cost effective and eco-friendly removal of lead from wastewater.
Mahzad Mirzaei; Reza Khanbabaie; Mohsen Jahanshahi; Ghasem Najafpour Darzi
Abstract
Recently, safety concerns over the handling of nanomaterials have become an important issue. The aim of the present study was to optimize the key parameters in the hydrothermal synthesis of CuInS2 quantum dots (QDs) as a non-toxic alternative to the cadmium-based QDs, that historically had dominated ...
Read More
Recently, safety concerns over the handling of nanomaterials have become an important issue. The aim of the present study was to optimize the key parameters in the hydrothermal synthesis of CuInS2 quantum dots (QDs) as a non-toxic alternative to the cadmium-based QDs, that historically had dominated the literature. Response surface methodology (RSM) in combination with D-optimal design was applied to optimize the synthesis and evaluate the PL intensity as the response which described by a reduced quadratic equation. The relationship between the PL intensity and independent variables (ligand/precursor, reaction time, reaction temperature, pH, and precursors ratio) was investigated using reduced quadratic polynomial equations. The produced QDs in the optimum condition were analyzed by UV-Vis, FE-SEM, and FTIR. The results showed that the nanoparticles have a high PL intensity and a red shift in both emission and absorbtion spectra which is a splendid point for their applications specially in bioimaging. The interaction between variables was not significant and the temperature was the most effective variable of PL intensity. A good agreement between predicted model and experimental data confirmed the correlated model.
