Asadollah Mohammadi; Parisa Abedi; Mohammad Reza Gholami
Abstract
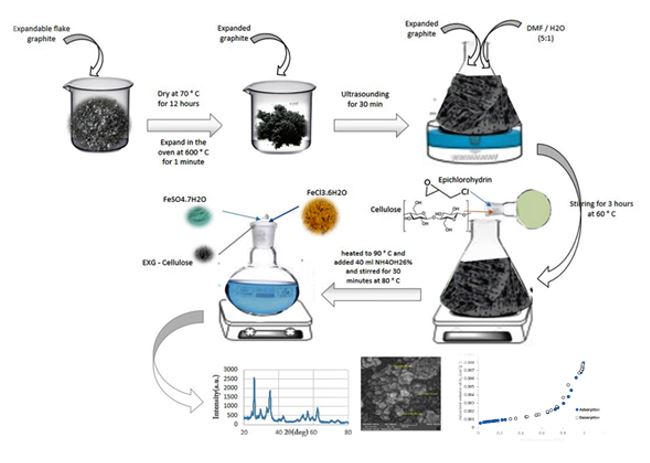
In this study, a new magnetic nanoadsorbent (denoted as Fe3O4/EXG/Cellulose) was synthesized using expanded graphite (EXG), microcrystalline cellulose and Fe3O4 nanoparticles. The Fe3O4/EXG/Cellulose was used as an effective nanoadsorbent for the removal of sulfamethoxazole (SMX) and malachite green ...
Read More
In this study, a new magnetic nanoadsorbent (denoted as Fe3O4/EXG/Cellulose) was synthesized using expanded graphite (EXG), microcrystalline cellulose and Fe3O4 nanoparticles. The Fe3O4/EXG/Cellulose was used as an effective nanoadsorbent for the removal of sulfamethoxazole (SMX) and malachite green (MG) from aqueous solutions using batch process. The Fe3O4/EXG/Cellulose was fully characterized by the Fourier transform infrared (FT-IR), X-ray diffraction (XRD), Energy-dispersive X-ray spectroscopy (EDX), Brunauer-Emmett-Teller (BET), Field emission scanning electron microscopy (FESEM), and transmission electron microscopy (TEM) analyses. Based on the BET analysis, an improved surface area from 10.32 to 71.86 m2 g–1 was achieved after the modification of expanded graphite using microcrystalline cellulose. The adsorption capacities for the MG dye and SMX drug were determined to be 109.9 and 3.9 mg/g, respectively. In addition, the adsorption process of SMX and MG by the Fe3O4/EXG/Cellulose was studied with isotherm and kinetic models. The results indicate the adsorption of SMX and MG by nanocomposite better described with pseudo-second-order kinetic. In addition, Langmuir and Freundlich isotherm models well describe the adsorption of MG and SMX using Fe3O4/EXG/Cellulose, respectively. Furthermore, Fe3O4/EXG/Cellulose can be easily recycled and reused for over 5 times and keeps a high level of adsorption efficiency.
Asadollah Mohammadi; Ali Aliakbarzadeh Karimi
Abstract
This study describes the adsorption and photocatalytic removal of methylene blue (MB) from aqueous solution by surface-modified TiO2 nanoparticles under ultraviolet irradiation in a batch system. The 5-sulfosalicylic acid grafted TiO2 (5-SA-TiO2) as a photocatalyst was characterized by means of ...
Read More
This study describes the adsorption and photocatalytic removal of methylene blue (MB) from aqueous solution by surface-modified TiO2 nanoparticles under ultraviolet irradiation in a batch system. The 5-sulfosalicylic acid grafted TiO2 (5-SA-TiO2) as a photocatalyst was characterized by means of XRD, FTIR and SEM techniques. The surface of TiO2 nanoparticles was modified by 5-sulfosalicylic acid (5-SA) to increase performance by altering surface properties. Notably, in contrast with the adsorption process, the remarkable removal enhancement of MB dye was observed by photocatalytic degradation process from aqueous solution. The adsorption and photocatalytic degradation kinetics of MB using 5-SA-TiO2 nanoparticles have also been investigated. The results show that the photocatalytic degradation was good fit with the pseudo-first-order kinetic model (R2 > 0.99). The adsorption isotherm of MB onto modified TiO2 nanoparticles fitted into the Temkin equation. In addition, thermodynamic studies indicate the spontaneous behavior of adsorption and photocatalytic degradation processes.
