Ghasem Zolfaghari
Abstract
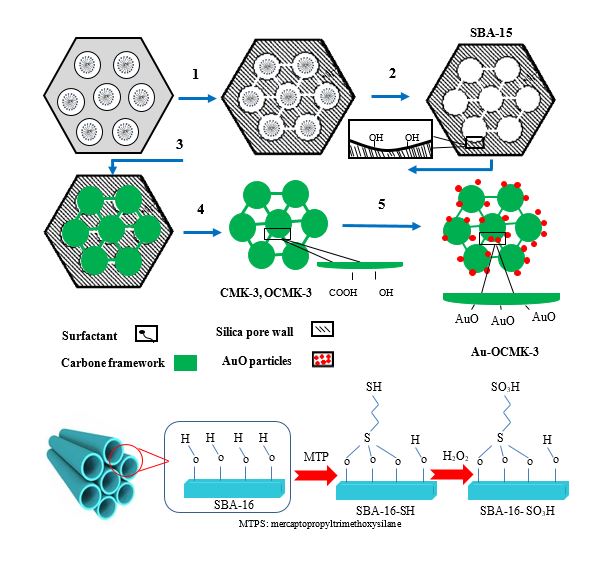
At present study, the removal of p-nitrophenol (PNP) by a newly designed mesoporous organocarbon, monolayers of ß-cyclodextrine (CD) on oxidzed ordered nanoporous carbon (OX-ONC) via 1,4-phenylene diisocyanate (PDI) linking denoted as CD-ONC was optimized. Furthermore, Au-doped mesoporous carbon ...
Read More
At present study, the removal of p-nitrophenol (PNP) by a newly designed mesoporous organocarbon, monolayers of ß-cyclodextrine (CD) on oxidzed ordered nanoporous carbon (OX-ONC) via 1,4-phenylene diisocyanate (PDI) linking denoted as CD-ONC was optimized. Furthermore, Au-doped mesoporous carbon CMK-3 denoted as Au-OCMK-3 was synthesized by using SBA-15. Au-OCMK-3 has been studied for removal of dibenzothiphene (DBT) and carbazole (CA) from n-hexane. Also the functionalization of SBA-16 mesoporous with sulfonic acid for arsenic (As (V)) and copper (Cu (II)) removal were carried out (SBA-16-SO3H). Maximum absorption capacity of CD-ONC was 100 mg/g. Dubinin–Radushkevich isotherm was applied to describe the nature of PNP uptake and it was found that it occurred physically (E = 0.07 KJ/mol, CD-ONC). Value for Temkin’s heat of adsorption is positive for PNP (157.87 J/mol, CD-ONC). There are two physisorption models of PNP with the surface C=O groups of ONC (H-bond and dispersion effect between phenolic ring and π electrons). The overall PNP adsorption process was exothermic and spontaneous in nature according to thermodynamics parameters (free energy (ΔGo), enthalpy (ΔHo), and entropy (ΔSo)). We demonstrate that functionalization of CMK-3 with gold is possible (qm value for DBT: 15.33 mg/g and for CA: 13.00 mg/g). The adsorption capacity for As (V) on SBA-16- SO3H reaches 92.63 mg/g. The high removal of As equilibrium time of 90 minutes can be explained in terms of a strong electrostatic attraction that occurred between the SO3H and As. Maximum absorption capacity was 92.63 mg/g for As(V) was and 13.00 mg/g for Cu(II).
kimia yadolahi; Arezoo Ghadi; Seyed Abolhasan Alavi
Abstract
The aim of this project is the production of The Descurainia Sophia adsorbent in nano dimensions using a super-grinding disk mill. The top-down method was used in the process of nanogel preparation. After complete drying of the stems, the stems were reduced to smaller sizes by an electric mill, and by ...
Read More
The aim of this project is the production of The Descurainia Sophia adsorbent in nano dimensions using a super-grinding disk mill. The top-down method was used in the process of nanogel preparation. After complete drying of the stems, the stems were reduced to smaller sizes by an electric mill, and by using the available sieve to standard number 60, adsorbent granulation was performed. The adsorbent was used to remove copper from aqueous solution. The tests and their optimization results were based on the design of experiments in three levels of variables using Taguchi method. According to the experiment results, pH, contact time, and adsorption mass are the main factors and the most influential effect on the removal of copper from the aqueous solution is the pH parameter, which has a lower p-value. As the pH increases from 5 to 9, the efficiency of copper metal removal increases. The concentration of hydrogen ions is the most important parameter affecting the adsorption process. The effect of temperature on adsorption efficiency has been investigated in the range of 20-50°C. Finally, the highest efficiency of copper cation removal was at 30°C and 8.89%. The results showed that the pH of the solution had the most effect on the copper removal efficiency and in the playing environment, the effect of copper removal was more than the acid and neutral conditions of the solution. Also, the adsorbent mass and contact time have the most effect on copper removal after the pH parameter in the Taguchi method.
Maryam Ghasemi; Somaye Mashhadi; Javad Azimi-Amin
Abstract
In this study, we have synthesized a new Fe3O4/AC nanocomposite using low cost adsorbent by microwave assisted in situ co-precipitation technique that was used as an effective adsorbent for the removal of methylene blue (MB) using the Taguchi design method as an optimization strategy. The optimum parameters ...
Read More
In this study, we have synthesized a new Fe3O4/AC nanocomposite using low cost adsorbent by microwave assisted in situ co-precipitation technique that was used as an effective adsorbent for the removal of methylene blue (MB) using the Taguchi design method as an optimization strategy. The optimum parameters are pH 7, Fe3O4/AC nanocomposite dose 0.03 g, contact time 30 min, initial concentration of MB 25 mg/L and temperature 298 K. The obtained results of ANOVA show that their percent contribution in descending order is pH (66.81%) > adsorbent dose (25.54%) > temperature (4.83%) > initial MB concentration (1.23%) > contact time (0.32%). The kinetic data were fitted to the pseudo-first-order, pseudo-second-order and intra-particle diffusion models and adsorption of MB dye followed pseudo-second-order kinetic. The obtained values of regression coefficient for Langmuir (0.98), Frendluich (0.93) and Dubinin–Radushkevich (0.94) showed that adsorption process fits to the Langmuir isotherm and the maximum adsorption capacity is 384.6 mg/g. Moreover, the thermodynamics studies suggested the spontaneous nature of adsorption process.
