Asadollah Mohammadi; Parisa Abedi; Mohammad Reza Gholami
Abstract
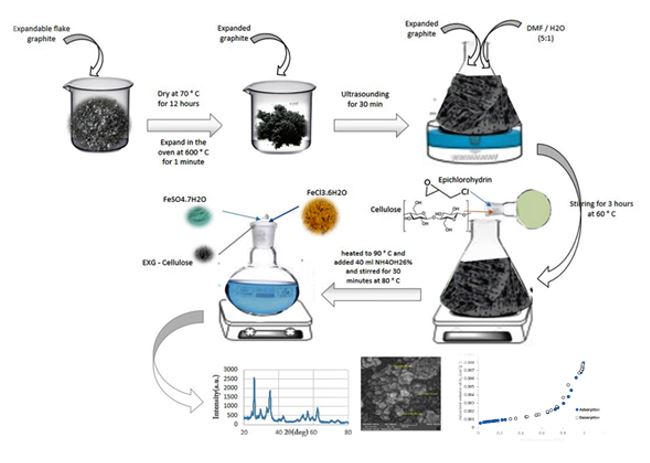
In this study, a new magnetic nanoadsorbent (denoted as Fe3O4/EXG/Cellulose) was synthesized using expanded graphite (EXG), microcrystalline cellulose and Fe3O4 nanoparticles. The Fe3O4/EXG/Cellulose was used as an effective nanoadsorbent for the removal of sulfamethoxazole (SMX) and malachite green ...
Read More
In this study, a new magnetic nanoadsorbent (denoted as Fe3O4/EXG/Cellulose) was synthesized using expanded graphite (EXG), microcrystalline cellulose and Fe3O4 nanoparticles. The Fe3O4/EXG/Cellulose was used as an effective nanoadsorbent for the removal of sulfamethoxazole (SMX) and malachite green (MG) from aqueous solutions using batch process. The Fe3O4/EXG/Cellulose was fully characterized by the Fourier transform infrared (FT-IR), X-ray diffraction (XRD), Energy-dispersive X-ray spectroscopy (EDX), Brunauer-Emmett-Teller (BET), Field emission scanning electron microscopy (FESEM), and transmission electron microscopy (TEM) analyses. Based on the BET analysis, an improved surface area from 10.32 to 71.86 m2 g–1 was achieved after the modification of expanded graphite using microcrystalline cellulose. The adsorption capacities for the MG dye and SMX drug were determined to be 109.9 and 3.9 mg/g, respectively. In addition, the adsorption process of SMX and MG by the Fe3O4/EXG/Cellulose was studied with isotherm and kinetic models. The results indicate the adsorption of SMX and MG by nanocomposite better described with pseudo-second-order kinetic. In addition, Langmuir and Freundlich isotherm models well describe the adsorption of MG and SMX using Fe3O4/EXG/Cellulose, respectively. Furthermore, Fe3O4/EXG/Cellulose can be easily recycled and reused for over 5 times and keeps a high level of adsorption efficiency.
Sumathi Harohally Paramesh; Veerendra Shetty Ananthpur; Nagaraju Rajendraprasad
Abstract
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were ...
Read More
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were effectively adsorbed onto NaOH activated LLPh (NaOH-LLPh) as bio-adsorbent. Employing the Chromatrap method within a column, successfully removed these dyes, while the surface morphology of the bio-adsorbent was elucidated through scanning electron microscopy (SEM) analysis. FTIR spectrometric data revealed valuable insights into the extent of adsorption. The impact of factors including adsorbate concentration, adsorbent dose, pH, contact time, and flow rate on the adsorption process was systematically studied and optimized. Up to 1000 µg/mL of MB and MG, 50 µg/mL of CV were found to be effectively removed by adsorption at pH 4-5, 3 and 2, respectively, at the flow rate of 1 mL/min. The results of kinetic studies and adsorption isotherms of above-mentioned dyes indicates that, all the three dyes follow the pseudo-second order kinetics. The adsorption of MB and MG are well fitted with the Langmuir isotherm model. The other dye CV suits with the Freundlich isotherm model. Based on the results, NaOH-LLPh, as an inexpensive and eco-friendly adsorbent, is suitable for the removal of cationic organic dyes from aqueous samples.

Reza Tayebee; vahid Mazruy
Abstract
The limited adsorption capacity of natural clays is a crucial and economic issue which confined their applications in industry as cheap adsorbents to remove toxic contaminants from wastewaters. Here, the adsorption capacity of a natural nano bentonite was enhanced by a simple acid and thermal activation ...
Read More
The limited adsorption capacity of natural clays is a crucial and economic issue which confined their applications in industry as cheap adsorbents to remove toxic contaminants from wastewaters. Here, the adsorption capacity of a natural nano bentonite was enhanced by a simple acid and thermal activation and the manufactured nano-adsorbent was characterized by FESEM, BET, FT-IR, and XRD. Effects of pH, temperature, sorbent capacity, and the initial concentration of malachite green were examined. The isotherm behavior of the adsorption system was investigated by the Langmuir and Freundlich isotherm models. Also, the kinetic inspections demonstrated that the adsorption of malachite green matched with the pseudo-second-order kinetic and the obtained thermodynamic parameters H, S, and G showed that the adsorption of malachite green was a spontaneous and endothermic process. The results indicated that the acid-thermal activated nano bentonite, with an enhanced surface area of >220 m2/g, can be depleted as a powerful and low-cost adsorbent to expel malachite green from aqueous solutions.

